Researchers cut up a 415 million year old fish fossil searching for the origin of the hardest-vertebrate tissue out there
Sometimes evolutionary concepts can be hard to come to terms with. Take for example, the fact that land vertebrates arose from fish.
This isn’t new information, but there’s a lot about this linkage that remains unknown, which is why researchers at Sweden’s Uppsala University and the Institute of Vertebrate Palaeontology and Palaeoanthropology (IVPP) in China are combining palaeontology and genomics in novel ways—hoping for more insight. In a September letter in Nature the team describes a bony-fish fossil from China that could reconfigure one part of the puzzle—the evolution of enamel.
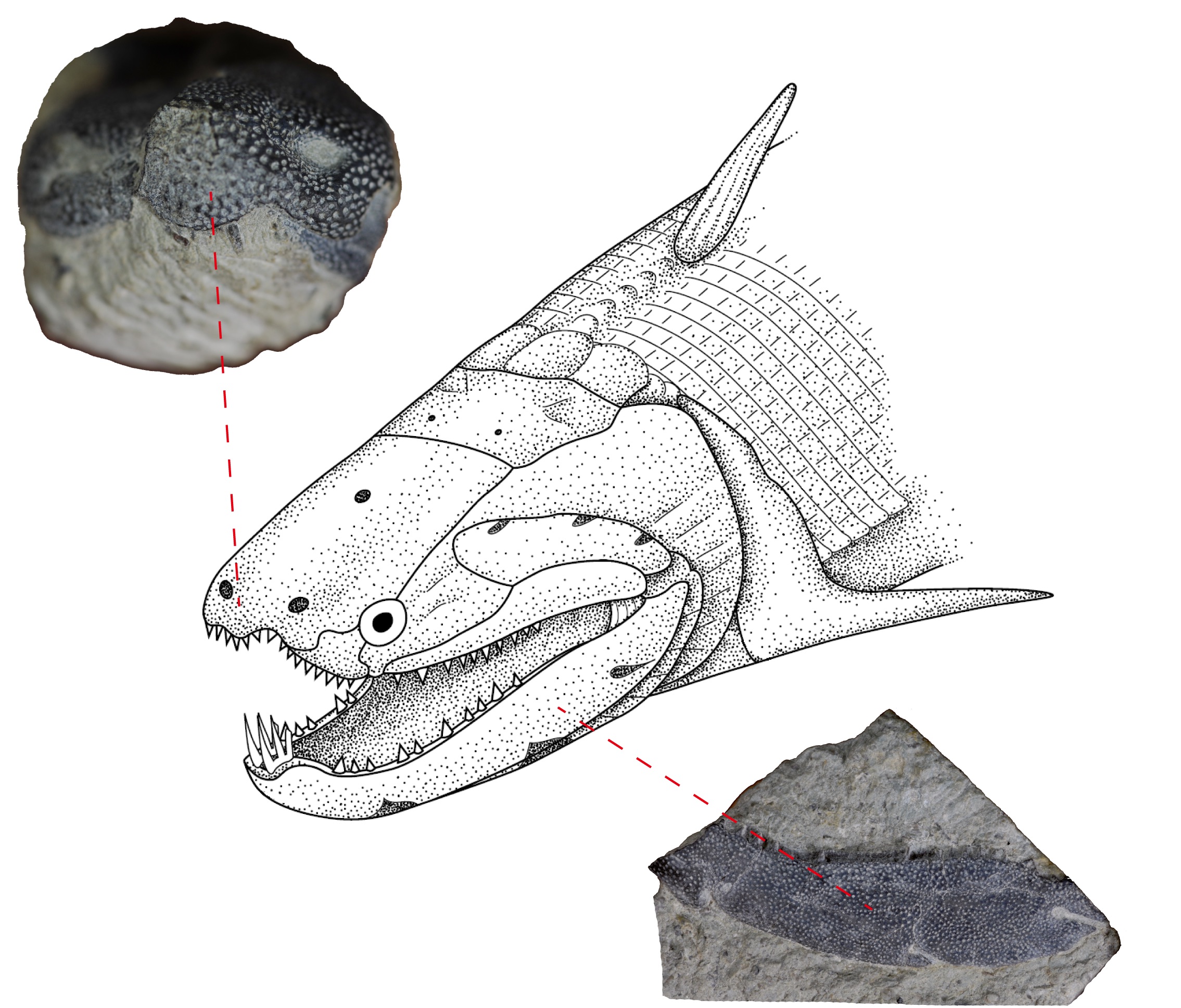
Most modern bony fish have enamel coating their dentine teeth like us, but some of their early ancestors are lacking it—despite possessing it on other regions of their bodies—making them great case studies. Psaroepis romeri is one such species, and unlike many ancient relics of life, there are quite a few specimens of P. romeri around. This emboldened their team to get physical with the fossil, explains letter co-author Per Ahlberg, a palaeontologist with Uppsala University.
Ahlberg has been personally studying early vertebrates for quite some time, and building up a diverse research team along the way. He says first authors Qingming Qu and Tatjana Haitina came to him with a plan to explore P. romeri, specifically to cut through the fossilised jawbone, skull and scales searching for the presence and boundaries of enamel.
‘We found the species had enamel on both, extending to within micrometres of naked dentine teeth,’ says Ahlberg.
Ahlberg says it’s already generally accepted that enamel had a dermal origin—based on the fact that the earliest enamel-bearers didn’t have jaws or teeth—but just how this happened is a mystery. Right now many theories hypothesise this occurred by migration, but these new findings lend credit to a different idea, that enamel instead switched up its boundaries.
Many animals in the past and today have enamel-patterning boundaries, places where the coating can and cannot be found, such as the torso, head or neck. Ahlberg says for some species, these borders are well defined, but with other species, like sharks, these boundaries are gone—with scaly-enamel their entire body length.
‘What we think now is that enamel switched its boundaries or spread across them,’ says Ahlberg. ‘We’re guessing this comes down to a change in gene enhancers.’
On that note, the team looked at the genome of a modern-day species, the Spotted gar (Lepisosteus oculatus), which has scale-coating enamel, and found genes that encode for two of the three fundamental enamel matrix proteins, crucial to the development of the hard coating. This is all exciting news, but Ahlberg says major questions still loam. How the dental system began, and how enamel came to cover teeth, are ultimately two separate questions with different timeframes that both need to be explored, he says. That’s why you need big, diverse teams working on queries of this nature, he adds.
‘What we’ve done here, coupling new material in both genomics and paleontology, is very rarely done, in fact this may be the first such example with early vertebrates,’ says Ahlberg. ‘That makes me especially proud of my team—whose success is the result of working together and thinking a bit differently.’

